Galvanic Cell vs Electrolytic Cell: A Guide for High School Chemistry
Many students find themselves confused when they first encounter the galvanic vs electrolytic cell comparison in electrochemistry. Both cell types involve redox reactions and electron transfer, yet they operate in fundamentally opposite ways. Once you understand the direction of energy flow and the role each electrode plays, the distinction becomes much clearer — and far less intimidating.
In this guide, HYE Tutors will walk you through what galvanic and electrolytic cells are, how each one works, the key differences between them, real-life examples you will recognise, and the common mistakes students make when studying this topic.
Galvanic vs Electrolytic Cell
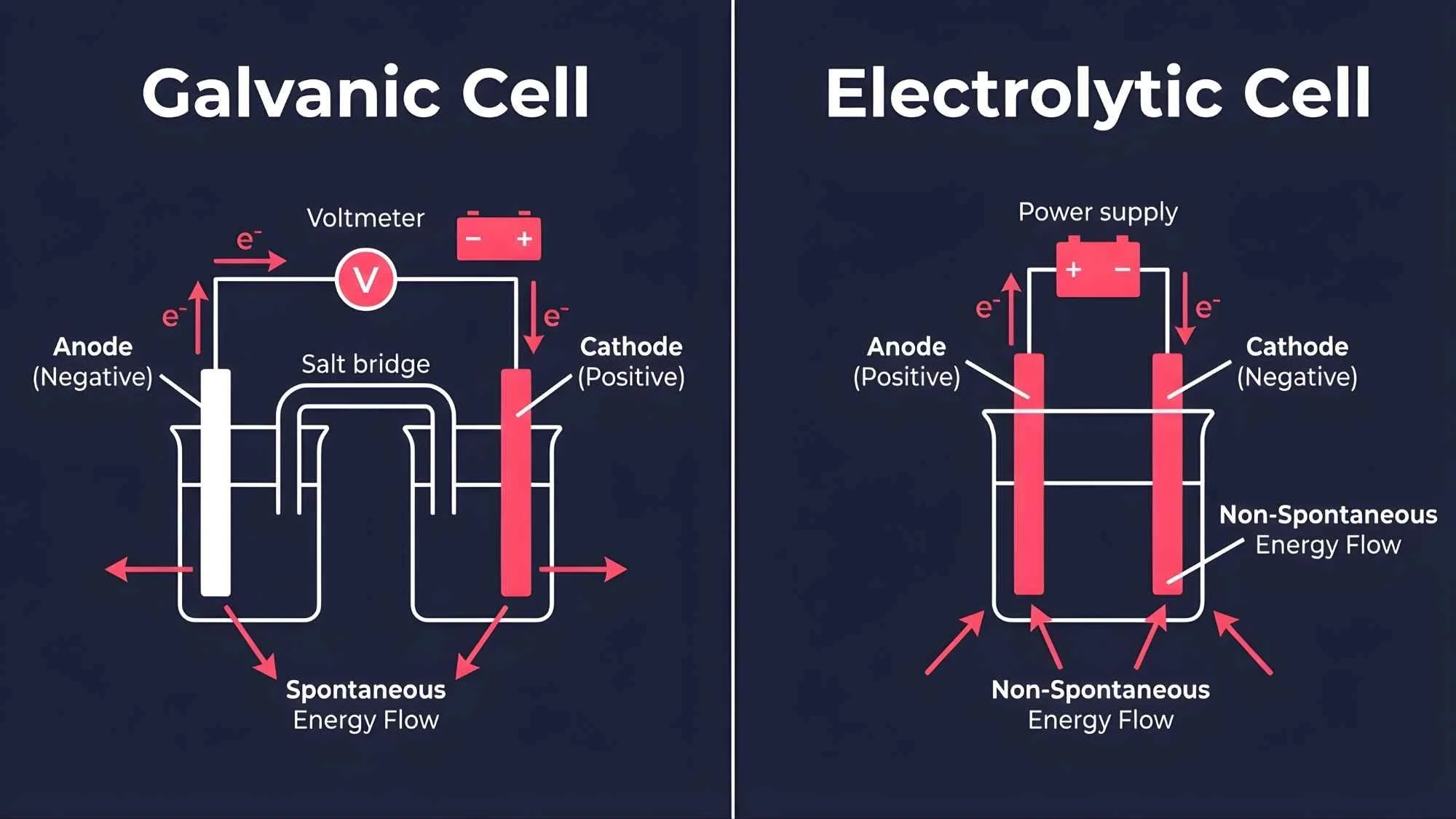
A galvanic cell (also called a voltaic cell) generates electrical energy from a spontaneous chemical reaction, converting chemical energy into electrical energy. An electrolytic cell does the opposite — it uses an external electrical supply to drive a non-spontaneous chemical reaction, converting electrical energy into chemical energy.
Galvanic cell → chemical energy to electrical energy (spontaneous)
Electrolytic cell → electrical energy to chemical energy (non-spontaneous)
Both systems rely on redox reactions and electron flow between two electrodes.
Galvanic Cell vs Electrolytic Cell
Before diving into each cell type, it helps to understand what they share. Both galvanic and electrolytic cells are electrochemical cells — systems in which chemical reactions produce or consume electrons. The critical difference is the direction of energy: one cell releases energy naturally, while the other needs energy pumped in from an outside source.
What Is a Galvanic Cell (Voltaic Cell)?
A galvanic cell, sometimes called a voltaic cell, converts chemical energy into electrical energy through a spontaneous redox reaction. Because the reaction happens on its own — without any external push — the cell produces a usable electric current. This is exactly how everyday batteries work.
Key features of a galvanic cell:
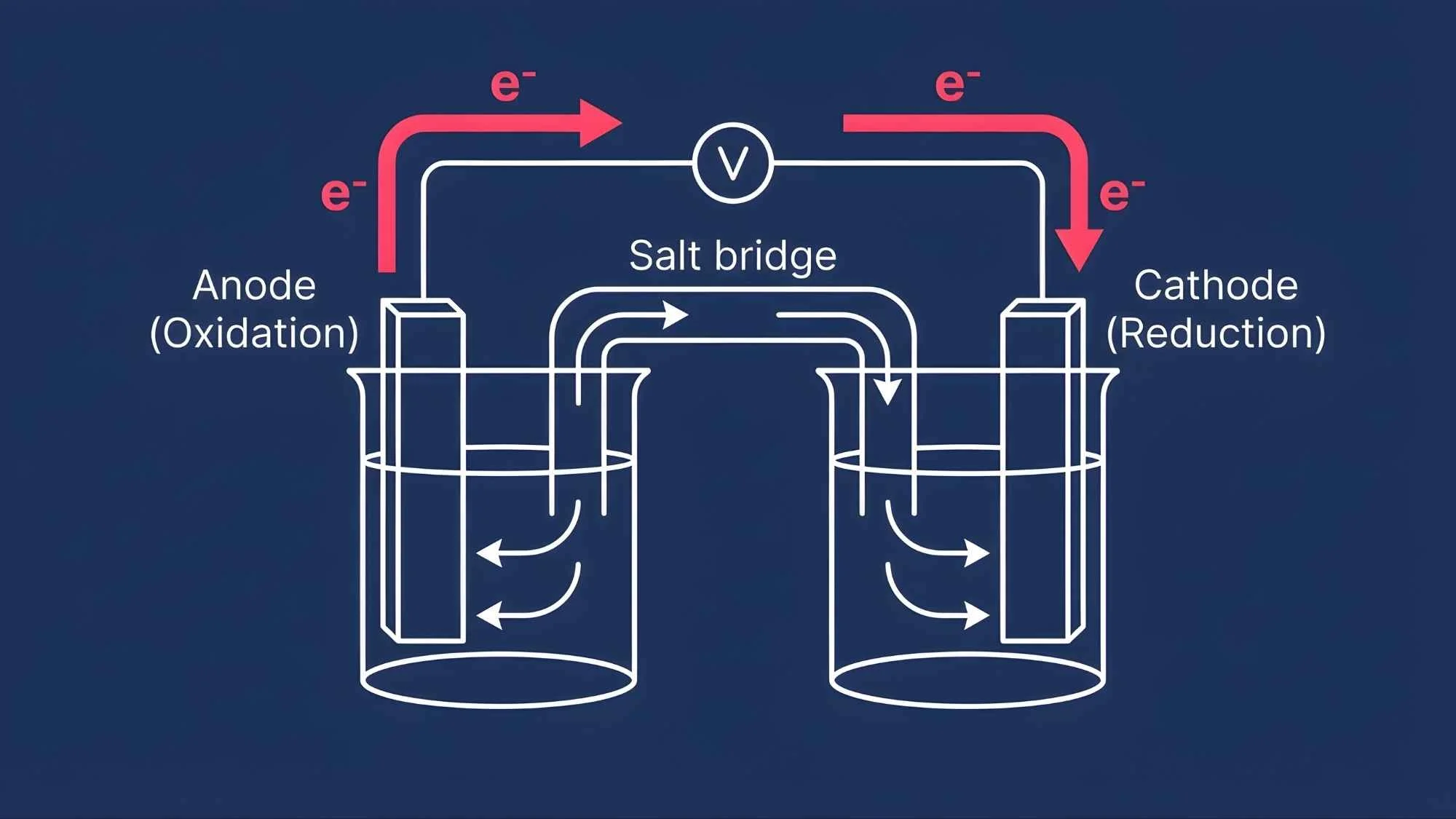
Oxidation takes place at the anode (the negative electrode), releasing electrons.
Reduction takes place at the cathode (the positive electrode), absorbing those electrons.
Electrons travel through an external circuit from anode to cathode, creating an electric current.
A salt bridge connects the two half-cells, maintaining electrical neutrality by allowing ion flow.
A classic textbook example is the Daniell cell, which uses a zinc anode and a copper cathode in separate electrolyte solutions. As Khan Academy explains, the spontaneous nature of the reaction is what makes galvanic cells so effective as portable power sources.
What Is an Electrolytic Cell?
An electrolytic cell works in reverse. Instead of generating electricity, it consumes electricity to force a chemical reaction that would not occur on its own — a non-spontaneous reaction. An external power supply pushes electrons through the system, driving oxidation and reduction at the electrodes.
Key features of an electrolytic cell:
Oxidation still occurs at the anode, but in an electrolytic cell the anode is connected to the positive terminal of the power supply.
Reduction still occurs at the cathode, which is connected to the negative terminal.
The external power supply provides the energy needed to drive the reaction forward.
No salt bridge is typically needed, as both electrodes are submerged in the same electrolyte solution.
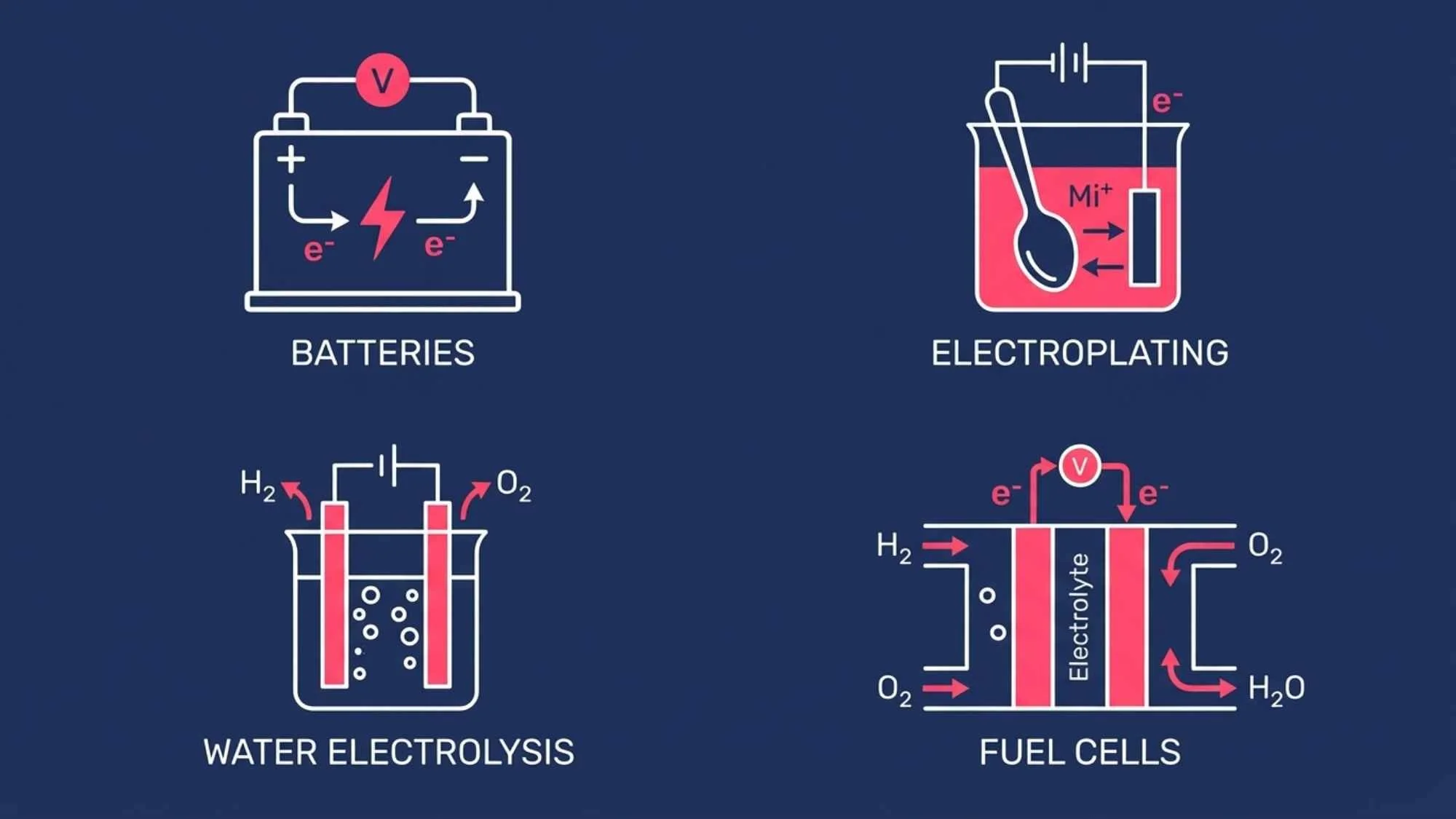
Common industrial applications include the electrolysis of water (splitting H₂O into hydrogen and oxygen gas) and electroplating, where a thin metal coating is deposited onto an object. According to LibreTexts Chemistry, electrolytic cells are fundamental to many large-scale industrial chemical processes.
Image 2
Key Differences Between Galvanic and Electrolytic Cells
The table below summarises every major point of comparison. Energy direction is the single most reliable way to keep the two straight in your mind.
| Feature | Galvanic Cell | Electrolytic Cell |
|---|---|---|
| Energy Conversion | Chemical → Electrical | Electrical → Chemical |
| Reaction Type | Spontaneous | Non-spontaneous |
| Power Source | None required | External electricity required |
| Anode | Negative electrode (oxidation) | Positive electrode (oxidation) |
| Cathode | Positive electrode (reduction) | Negative electrode (reduction) |
| Energy Flow | Releases energy | Consumes energy |
| Common Uses | Batteries, fuel cells | Electroplating, electrolysis |
| Example | Daniell cell, AA battery | Electrolysis of water, electroplating |
How Electron Flow Works in Electrochemical Cells
Electron flow is the heartbeat of any electrochemical cell. Understanding where electrons come from and where they go makes the whole topic click into place.
Here is the process broken down conceptually:
Oxidation at the anode: The anode material loses electrons during oxidation. These electrons become available to travel through the external circuit.
External circuit travel: Electrons flow from the anode, through the wire (and any connected device), to the cathode. This movement of electrons is the electric current.
Reduction at the cathode: At the cathode, those electrons are absorbed by ions or molecules in a reduction reaction.
One of the most common points of confusion is electrode charge. In a galvanic cell, the anode is negative and the cathode is positive — because the anode pushes electrons out. In an electrolytic cell, the power supply reverses the apparent charge: the anode is connected to the positive terminal and the cathode to the negative terminal. Despite this, the underlying rule never changes:
| Electrode | Process | Galvanic Charge | Electrolytic Charge |
|---|---|---|---|
| Anode | Oxidation (loses e⁻) | Negative | Positive |
| Cathode | Reduction (gains e⁻) | Positive | Negative |
As the Royal Society of Chemistry notes, keeping the definitions of anode (oxidation) and cathode (reduction) firmly in mind is the foundation of understanding any electrochemical system.
Real-Life Examples of Galvanic and Electrolytic Cells
Electrochemistry is all around us. Once you know what to look for, you will spot both cell types in everyday life and industry.
Examples of Galvanic Cells
AA and AAA batteries: The most familiar galvanic cells, converting stored chemical energy into electrical energy to power devices.
Lithium-ion batteries: Found in smartphones and laptops, these are advanced galvanic cells that can also be recharged (making them rechargeable galvanic systems).
Fuel cells: Used in electric vehicles and space technology, fuel cells continuously convert hydrogen and oxygen into electricity through spontaneous redox reactions.
Examples of Electrolytic Cells
Electroplating jewellery and cutlery: A thin layer of silver or gold is deposited onto a base metal using an electrolytic cell — the object to be plated acts as the cathode.
Aluminium extraction: Aluminium is produced industrially from aluminium oxide (bauxite) using large-scale electrolytic cells, a process known as the Hall–Héroult process.
Electrolysis of water: Passing an electric current through water splits it into hydrogen gas at the cathode and oxygen gas at the anode — a key process in green hydrogen energy production.
Electrochemistry sits at the intersection of energy, materials science, and biology, making it one of the most applied areas of high school and university chemistry.
Common Mistakes Students Make When Comparing These Cells
As a chemistry tutor, these are the errors I see students make most often — and they are all easy to fix once you are aware of them.
Mixing up energy direction: This is by far the most common mistake. Remember — galvanic cells generate electricity; electrolytic cells require it. When in doubt, ask: where is the energy coming from?
Confusing anode and cathode charges: Students often remember that the anode is negative in a galvanic cell and accidentally apply that to electrolytic cells too. In electrolytic cells, the anode is connected to the positive terminal of the power supply — though oxidation still occurs there.
Assuming both cells produce electricity: Only galvanic cells produce electricity. Electrolytic cells consume it.
Misunderstanding spontaneous vs non-spontaneous: A spontaneous reaction releases free energy on its own. A non-spontaneous reaction needs energy input. Galvanic cells harness spontaneous reactions; electrolytic cells force non-spontaneous ones.
Keep coming back to the golden rules: Galvanic = Generates. Electrolytic = Requires electricity. Anode = Oxidation. Cathode = Reduction. Every time.
FAQs
What is the difference between galvanic and electrolytic cells?
A galvanic cell converts chemical energy into electrical energy through a spontaneous redox reaction — no external power is needed. An electrolytic cell uses external electrical energy to drive a non-spontaneous chemical reaction. The key difference is the direction of energy: galvanic cells produce electricity, while electrolytic cells consume it.
Which cell produces electricity: galvanic or electrolytic?
A galvanic cell produces electricity. It harnesses a spontaneous chemical reaction to generate an electric current, which is why everyday batteries are galvanic cells. An electrolytic cell does the opposite — it requires an external electricity supply to make a chemical reaction happen.
Why do electrolytic cells require an external power source?
Electrolytic cells drive chemical reactions that are non-spontaneous, meaning they do not occur naturally on their own. An external power source — such as a battery or mains electricity — provides the energy needed to push the reaction forward, forcing oxidation and reduction at the respective electrodes.
What are real examples of galvanic and electrolytic cells?
Galvanic cell examples include AA batteries, lithium-ion phone batteries, and hydrogen fuel cells. Electrolytic cell examples include the electroplating of jewellery, industrial aluminium extraction (the Hall–Héroult process), and the electrolysis of water to produce hydrogen and oxygen gas.
Conclusion
Understanding the galvanic vs electrolytic cell distinction is one of the most important building blocks in high school electrochemistry. At their core, these two cell types are mirror images of each other: one releases energy from a spontaneous reaction to produce electricity, and the other uses electricity to force a non-spontaneous reaction to occur.
The easiest way to keep them straight is always energy direction. Galvanic cells generate electrical energy; electrolytic cells consume it. Pair that with the firm rule that oxidation always happens at the anode and reduction always happens at the cathode, and you have the foundation of electrochemistry covered.
With clear explanations, good comparison tables, and plenty of practice, mastering electrochemistry concepts is absolutely achievable — and HYE Tutors is here to help every step of the way.