How to Find Limiting Reactant from Balanced Equation
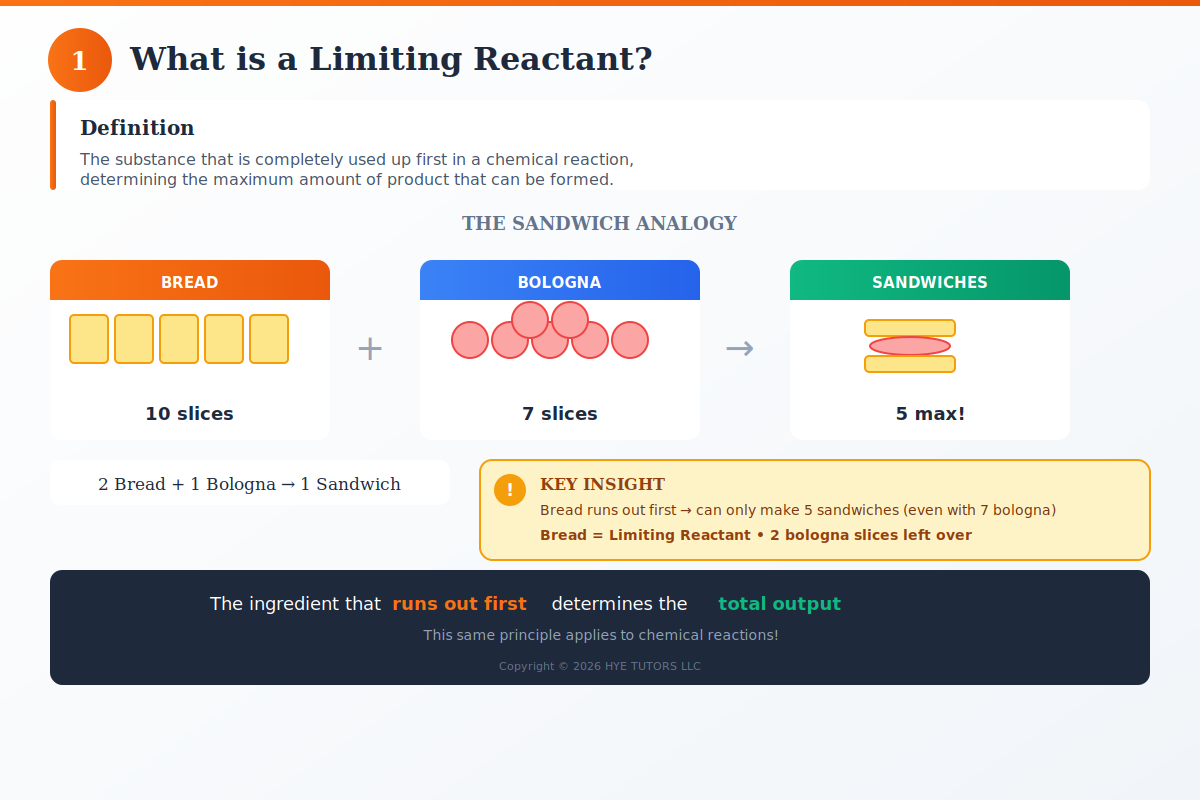
In chemistry, the limiting reactant is the substance that gets completely used up first, stopping the reaction from continuing. Think of it like running out of bread when making sandwiches—even if you have plenty of cheese and meat, no bread means no more sandwiches. The limiting reactant controls the maximum amount of product your reaction can produce, which is why understanding how to find the limiting reactant is essential for stoichiometry success.
“The limiting reactant is the substance that runs out first in a chemical reaction, determining how much product can be made. You can find it in four easy steps: (1) balance the equation, (2) convert all reactant amounts to moles, (3) divide each amount by its coefficient to find the mole ratio, and (4) identify the reactant with the smallest ratio — that’s your limiting reactant.”
How to Find Limiting Reactant from Balanced Equation
Every method for finding the limiting reactant starts with a balanced chemical equation. The coefficients in front of each substance tell you the exact ratios in which reactants combine and products form. Without this foundation, your calculations won't reflect reality.
Step 1: Write the Balanced Chemical Equation
Before anything else, make sure your equation is balanced. This means the number of atoms of each element must be equal on both sides of the arrow. Only adjust coefficients (the numbers in front of formulas)—never change the subscripts within chemical formulas.
Why does this matter? Unbalanced equations give you incorrect mole ratios, leading to wrong answers. If your equation shows 2H₂ + O₂ → 2H₂O, those coefficients (2, 1, and 2) are the key to everything that follows. For a refresher on balancing equations, the Khan Academy chemistry course offers excellent practice problems.
Step 2: Convert All Reactants to Moles
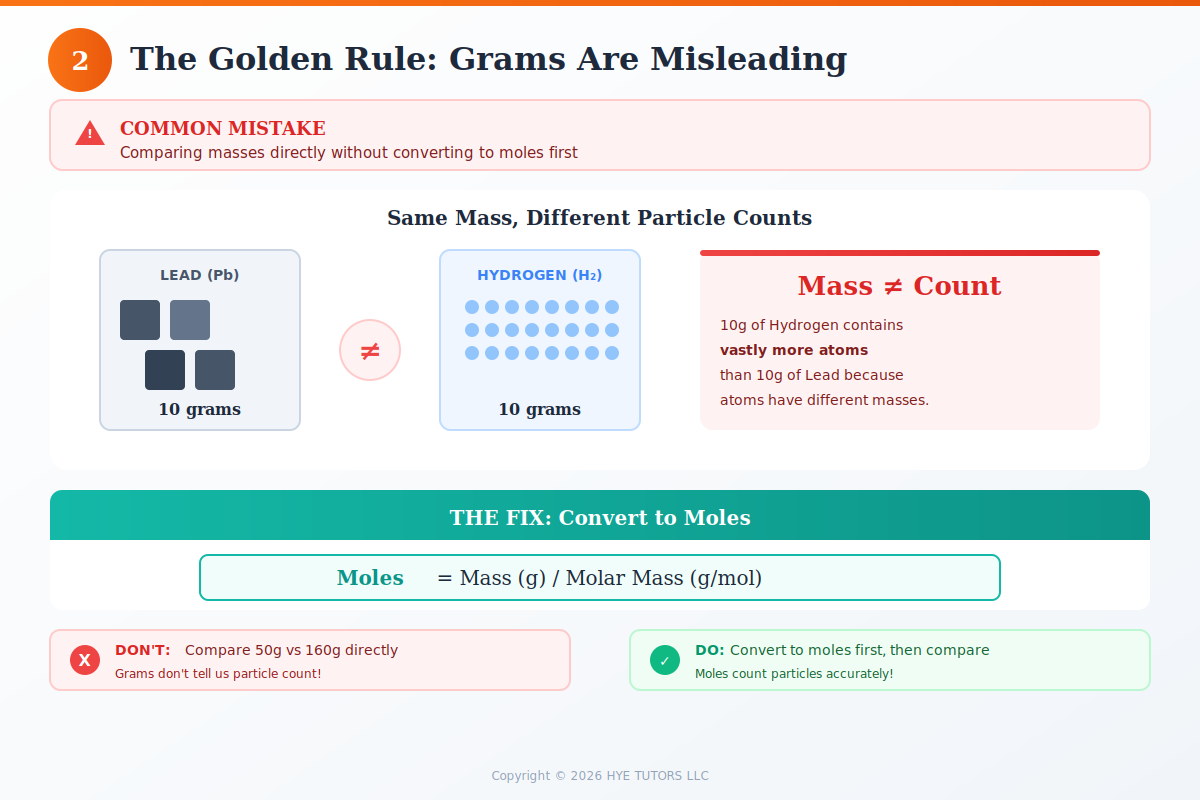
You can't compare grams of different substances directly because atoms have different masses. Converting to moles puts everything in the same "language." Use the formula:
moles = mass (g) ÷ molar mass (g/mol)
Find molar masses using the periodic table by adding up the atomic masses of all atoms in each formula. This step is non-negotiable—comparing grams instead of moles is one of the most common mistakes students make.
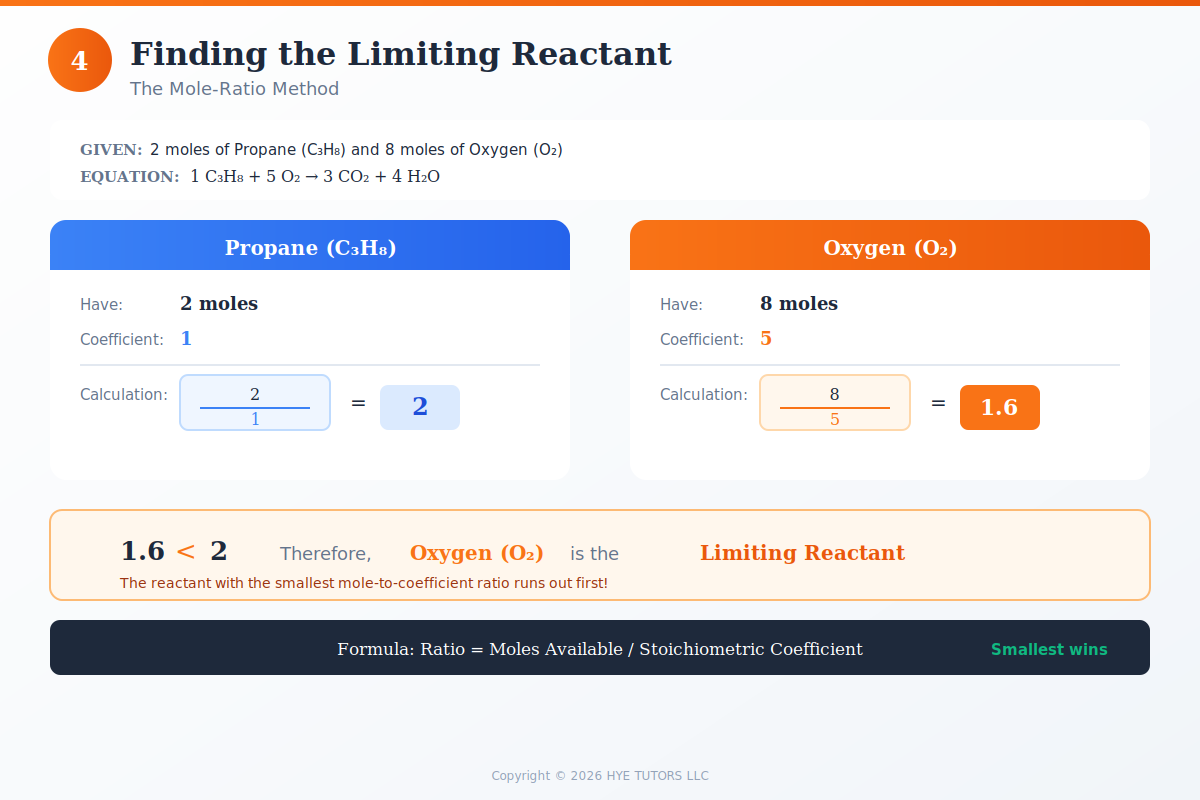
Step 3: Compare Mole Ratios Using the Balanced Equation
Now comes the critical comparison. Divide the moles of each reactant by its coefficient from the balanced equation. This tells you how many "complete sets" of the reaction each reactant can support.
The reactant with the smallest ratio is your limiting reactant. Why? Because it will run out first, stopping the reaction before the other reactants are used up. This is how to find the limiting reactant with moles—by comparing what you have to what the reaction requires.
Step 4: Identify the Limiting Reactant and Confirm
To double-check your answer, calculate how much product each reactant would produce if it were the limiting one. The reactant that yields the smaller amount of product is confirmed as the limiting reactant. This verification step also reveals which reactant is in excess—it's simply whichever one isn't limiting.
How to Find Limiting and Excess Reactant
Once you've identified the limiting reactant, everything else is the excess reactant—the substance that remains after the reaction completes. To calculate how much excess reactant is left over, first determine how much of it reacted (using stoichiometry from your balanced equation), then subtract that from what you started with.
Understanding how to find limiting and excess reactant matters beyond the classroom. In laboratories and industrial processes, minimizing excess reactants reduces waste and saves money. Pharmaceutical companies, for example, carefully calculate reactant quantities to maximize efficiency and minimize costly leftover materials.
How to Find the Limiting Reactant with Moles
Sometimes problems give you moles directly rather than grams. This actually simplifies the process—you skip the mass-to-moles conversion entirely. Start with your mole values, divide each by its coefficient from the balanced equation, and compare. The reactant with the smallest result is limiting.
This "mole-ratio method" is particularly useful when working with solutions (where concentration × volume gives you moles) or gas law problems. The core logic remains identical: whoever runs out first determines how much product you get.
How to Find Limiting and Excess Reactants: Worked Example
Problem: You have 4 moles of hydrogen (H₂) and 3 moles of oxygen (O₂). Using the reaction 2H₂ + O₂ → 2H₂O, find the limiting reactant.
Solution: Divide each reactant's moles by its coefficient. For hydrogen: 4 mol ÷ 2 = 2. For oxygen: 3 mol ÷ 1 = 3. Since hydrogen has the smaller ratio (2 < 3), hydrogen is the limiting reactant. Oxygen is in excess.
Check: Using 4 mol H₂ produces 4 mol H₂O. Using 3 mol O₂ would produce 6 mol H₂O. Since 4 mol < 6 mol, hydrogen limits the reaction. We'll make 4 moles of water, with 1 mole of O₂ left over.
Common Mistakes Students Make When Finding the Limiting Reactant
Forgetting to balance the equation: An unbalanced equation gives incorrect ratios. Always verify both sides have equal atoms before proceeding.
Comparing grams instead of moles: 10 grams of hydrogen is very different from 10 grams of oxygen. Moles, not mass, determine reactant ratios.
Ignoring coefficients: Simply comparing moles without dividing by coefficients leads to wrong answers. The coefficients tell you how much of each substance the reaction actually needs.
Rounding too early: Keep extra decimal places throughout your calculation and round only at the final answer. Early rounding compounds errors. The ChemLibreTexts stoichiometry resources provide additional practice with proper significant figures.
FAQs
What is a limiting reactant in chemistry?
A limiting reactant is the substance that gets completely consumed first in a chemical reaction, determining the maximum amount of product that can form.
Why do we need to find the limiting reactant?
Finding the limiting reactant tells you how much product a reaction will actually produce and helps you plan efficient use of materials in labs and industry.
Can there be two limiting reactants?
Only in the rare case where two reactants would be completely consumed at exactly the same moment—this happens when they're present in perfect stoichiometric ratios.
Is limiting reactant the same as theoretical yield?
No—the limiting reactant is a substance, while theoretical yield is the amount of product calculated from the limiting reactant.
How do I know which reactant is in excess?
After identifying the limiting reactant, all other reactants are in excess. You can calculate how much excess remains by subtracting what reacted from your starting amount.
Wrap Up
Here's your go-to checklist for how to find limiting reactant chemistry problems: First, write and balance your chemical equation. Second, convert all given amounts to moles using molar mass. Third, divide each reactant's moles by its coefficient. Fourth, identify the smallest ratio—that's your limiting reactant. The other reactant(s) are in excess. For extra confidence, verify by calculating theoretical yield from each reactant.
Need more help mastering stoichiometry? At HYE Tutors, we specialize in making chemistry click for students at every level. Our tutors break down complex concepts into manageable steps—just like we did here. Reach out today to build confidence in your chemistry skills.